Medical devices
Millions of patients
Treated with our technology in numerous regulatory-cleared Class II and Class III devices.
$25 billion revenue
Generated for leading medical device multi-national companies, start-ups, and venture firms.
10,000 procedures
Of deep clinical, workflow, and procedure insights that drive our innovations.
Hundreds of clinicians
Collaborate with our R&D teams across hospitals in US, EU, and APAC.
What we do
We tackle the world’s toughest healthcare challenges, developing novel products, platforms and ecosystems to improve outcomes, care delivery, patient experience, and healthcare value. We have deep experience developing medical devices across a range of clinical applications.
Smart Implants
As the largest independent smart implant development firm, we develop highly novel implant systems, closed-loop control therapy, and digital ecosystems to enable a new generation of smart implants to address the most challenging disease states.
We’ve worked on over 3 dozen implant programs over the past 20 years, with multiple FDA clearances and CE markings. Our implant experience includes deep brain stimulation, vagal nerve, spinal cord, urology, orthopedics, cochlear and auditory, cardiovascular, cardiac rhythm management, pancreatic, and ophthalmic applications.
We specialize in implant system development, novel sensing, tissue-device interface, closed-loop control therapy, safety-critical software, implant power, communication, data security, artificial intelligence, real-world evidence, and digital ecosystems.
Robotics & Visualization
Robotic, endoscopic, and laparoscopic systems are expanding into exciting new clinical applications, enabling procedure innovation for minimally invasive surgery. We partner with ambitious clients to conceptualize, design, and deliver the advanced surgical systems of tomorrow.
Over two decades our teams have been involved with many robotic developments, spanning robotic system architecture, robotic instrument development, safety and control architecture, surgical navigation, advanced optics, novel sensing, and human factors engineering.
Our advanced visualization expertise includes fluorescence imaging, near-infrared, multi-spectral and hyperspectral imaging, ultrasound, OCT, meta-surfaces, nanofabrication, digital holography, integrated photonics and quantum sensing.
Digital Surgery
Digital Surgery can dramatically improve the standard of operative care for patients worldwide. We develop digital surgery platforms and ecosystems that augment surgeon decision-making, reduce outcomes variability, and increase procedural efficiency to improve the standard of patient care.
We architect high value digital surgery platforms by combing deep expertise in robotics, advanced visualization, sensing, optics, artificial intelligence and machine learning, augmented reality, human factors, human machine understanding, wireless connectivity, digital service design, and business model transformation.
Critical Care
Critical care medicine can benefit from high value digital ecosystems to streamline care, reduce provider burden, and improve patient outcomes. We accelerate integration of break-through sensing technology, AI, algorithms, and connectivity into critical care devices and platforms.
Critical care is a highly connected data rich environment with many opportunities for “intelligent assist” systems incorporating digital technologies to augment the capabilities of healthcare professionals and support the care process.
We bring a deep understanding all enabling technologies along the signal chain from sensing to security and can help solve complex, multidisciplinary problems. We apply this in the context of system engineering, workflow, human centred design and patient-device interaction.
Featured insights
Our expertise
Cardiovascular disease is ripe for technology disruption. We develop advanced cardiovascular implants, tissue sensing, biomarkers monitors, minimally invasive devices, and cross-fertilize medtech innovation into cardiovascular applications.
Our teams have worked on Class III implants and advanced interventional systems in pacing, defibrillation, electrophysiology, structural heart, peripheral vascular indications.
We have extensive experience in advanced laparoscopy, next-generation robotic platforms, robotic instrumentation, and novel flexible endoscopy for colo-rectal, gastro-intestinal, bariatric, pancreatic, urologic, and general abdominal procedures.
Our teams are deploying cutting-edge technology to address lung cancer globally. We’ve been active in advanced molecular diagnostics, intra-operative sensing, novel energy therapy, drug-device therapy, and bronchoscopy platforms. Our mission is to help industry leaders develop holistic integrated solutions for lung cancer patients.
We invent technology and develop ground-breaking products addressing Parkinson’s Disease, epilepsy, multiple sclerosis, traumatic brain injury, sleep apnea, incontinence and pain management.
We are innovation partners to the North America Neuromodulation Society, partners with leading multi-national companies and dynamic startups, and collaborate with leading neurosurgeons and Harvard University to drive clinical and scientific innovation.
Our ophthalmology innovation teams have decades of experience working on platforms to address age-related macular degeneration, glaucoma, diabetic retinopathy, working on intraocular lenses, optical coherence tomography, phaco-emulsification, and surgical robotic and AI platforms. We regularly partner with leading KOL’s, hospitals, and industry forums like OIS to advance eye care.
We played a large role in the orthopedic industry’s technology evolution. We’ve developed a range of enabling technologies to transition high volume procedures in knees, hips, spine, shoulder, and trauma to capture greater value for patients, hospitals, and industry leaders. We have significant experience in surgical navigation, surgical robotics, digital surgery platforms, and implantable sensors for orthopedic procedures.
Our mission is to deliver transformational innovation to impact women’s health globally. We develop innovative products, technology, and platforms addressing significant unmet needs in maternal care, reproductive health, breast cancer and cardiovascular disease.
We partner with leading multi-national companies, clinicians, and visionary startups to drive actionable change across industry, clinical practice, and patient care. We are proud to lead Women’s Health initiatives such as our recent Women in Medtech panel in partnership with MassMEDIC.
Our work
Collaborations and partnerships
















Our facilities
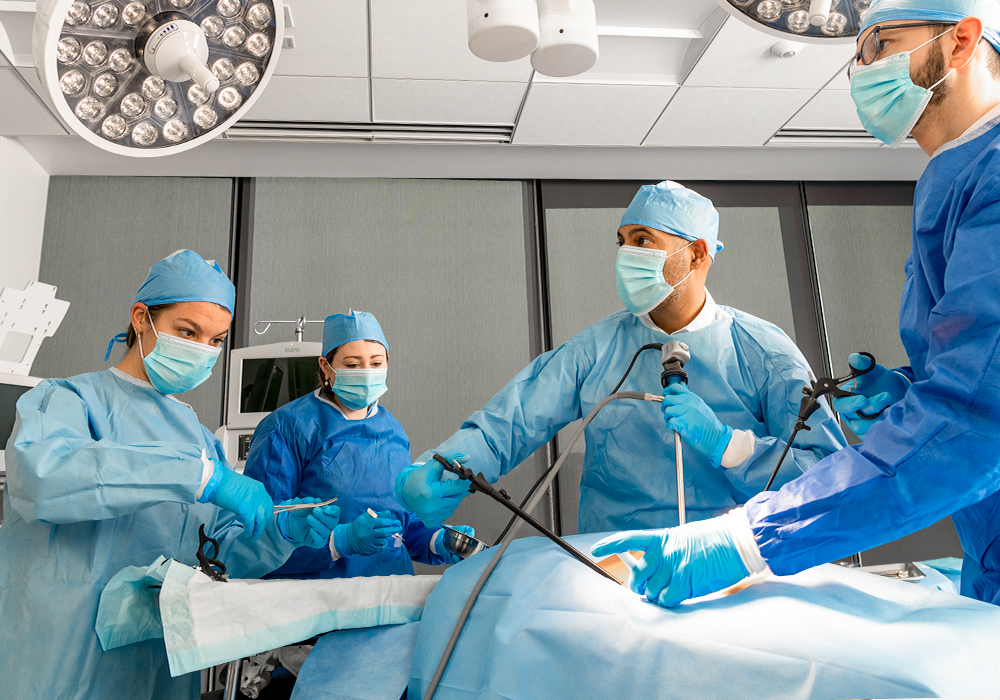 |
Digital operating roomOur digital operating room is a unique innovation lab enabling clients and surgeons to collaborate with our R&D teams on transforming the future of surgery. This innovation space accelerates the development of digital surgery platforms, such as next-generation surgical robots and visualization systems, as well as smart implants and critical care systems. Clients use our digital operating room for strategy and visioning workshops, human factors formative research, concept brainstorming, prototype development and verification testing of complex digital surgery platforms. |
 |
Human factors labThis human factors Workshop is a hub for user-centred design. Our consultants can meet a variety of client and project needs in the highly adaptable space, such as creating a comfortable home environment for a patient, hosting large client teams for observation of a simulated surgery or discussing strategic product visions with a panel of stakeholders. Here we conduct user research and obtain product feedback quickly and effectively, keeping users at the forefront of design. |
 |
Anechoic test chamberOur custom anechoic radio and antenna test chamber is a highly sophisticated 5m cube. Inside, we carry out automated 3D measurements and visualisation of wireless device behaviour and test body-worn technology using body phantoms or real live test subjects to optimise real-word RF performance of products such as smart implants (including implanted telemetry), connected drug delivery devices and wearables. Our anechoic test chamber is available as a standalone service for industry and academic research. Explore the facility and find out how you can access this state-of-the-art service. |
 |
AI infrastructureCC’s comprehensive onsite compute infrastructure allows us to explore options and train and test models more rapidly to get better results. We run petaflop-scale, containerised compute on site, with multiple NVIDIA DGX-1 deep learning supercomputers and other GPU and FPGA-accelerated servers. This links to petabyte-scale local NetApp storage, project-specific cloud and our continuous integration systems. |
Our experts

Rahul Sathe
Senior Vice President - Medtech Innovation

John Genova
Senior Vice President, Commercial Head Smart Implants

Ashley Hawson
Senior Vice President Global Commercial Operations

Mike Hobby
Associate Director of Medical Devices and Life Sciences

Alex Maret
Vice President, Robotics & Digital Surgery

Maureen Halligan
Vice President, Advisory & Innovation Services





